
A new application for an old drug: How does ketamine battle depression?
According to the researchers from three Canadian universities, a group of proteins known as 4E-BPs, involved in memory formation, is the key to unlocking the antidepressant effect of ketamine in the brain. The discovery could lead to more acceptable and safer treatments for specific patients suffering from major depression.
It is challenging to find an effective treatment for the major depressive disorder because more than 30% of patients are resistant to selective serotonin reuptake inhibitors (SSRI), the most commonly-prescribed antidepressants.
Ketamine for Perioperative Pain Management
Initially, researchers approved ketamine for anesthesia and pain relief. Following its discovery, researchers have been studying new uses for this drug, and ketamine was approved last year for patients suffering from major depression who are treatment-resistant. Ketamine works within hours, unlike standard antidepressants, which can take several weeks to show an effect. Until now, the information about the molecular mechanism that triggers the antidepressant effect of ketamine on the brain was minimal.
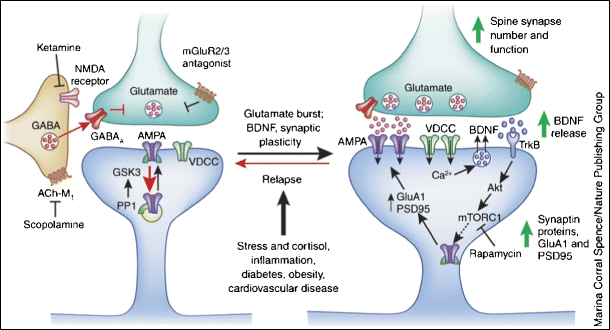
A study published in Nature by researchers from McGill University, Université de Montréal, and Carleton University studied the effect of ketamine on response and neuronal activity in mice. Using genetic tools to remove proteins from specific brain cells, the team found that when 4E-BPs are absent in the brain, particularly in neurons, ketamine cannot produce its antidepressant effect. 4E-BPs works as a switch to turn on or off the process of protein synthesis—an indispensable component of memory formation.
How does ketamine combat depression?
“This is an excellent instance of how basic research, in this case, the control of protein synthesis, drives to major discoveries in understanding disease, and the hope of curing it,” says co-author Nahum Sonenberg, a professor at the Department of Biochemistry at McGill University.
The researchers investigated the role of 4E-BPs on ketamine’s effect on two major types of neurons:
One is the excitatory neurons, which make up most neurons in certain parts of the brain. The other one is the inhibitory neurons, which control excitatory neurons and significantly affect behavior.
“We were hoping that 4E-BPs would only be relevant in excitatory cells, but surprisingly, eliminating 4E-BPs from inhibitory cells was sufficient to block the effect of ketamine,” says co-author Jean-Claude Lacaille, a professor at the Department of Neurosciences at Université de Montréal.
Medicine is not one-size-fits-all.
The development and approval of ketamine for treatment-resistant patients were considered a significant advance in modern psychiatry. Despite its promise, ketamine remains a less-than-perfect therapy because it can be addictive. The researchers hope that their findings will pave the way for better and safer antidepressant treatments for primary depressive disorder patients.
“More delay can prolong the suffering of patients and affect their quality of life,” says co-author Aguilar-Valles, a former research associate at McGill University and presently an assistant professor at Carleton University. “Our discovery has the potential to draw us closer in finding a safer alternative to ketamine, and ultimately to a personalized medicine approach.”
The study also included a clinician-researcher, Dr. Gabriella Gobbi, from the Department of Psychiatry at McGill University, who works with individuals affected with depression and other psychiatric illnesses.
In the next steps, the researchers will examine whether males and females have different responses to ketamine. It could have important implications for treatment for people with depressive disorders, which significantly overrepresents women.
good information
At last, someone wrote something very important about such a hot topic and it is very relevant nowadays.
best place to buy pills in the USA.
How do you get a response after buying them?
Nothing to add, just I love to buy medicine on this website and read every word here.
I this like as well:
I find all the meds that I need in one place at buyambienpills.com
Medicines are cheaper here compared to other websites I recommend this store.
overnight delivery is best when you need medicine in an emergency and they are in good.
I ordered this medicine and got it shipped overnight.
I selected overnight delivery but there was a delay and it took more than 2 days to get my meds.
I got a heavy discount by paying using bitcoin.
They provide fedex delivery overnight.
Buying Percocet on this site at a discount rate is really amazing. Thank Purdue Pharm
These people have both brand and generic medicines
Medicines are cheaper here compared to other websites.
Best pain medicines are avaialbe here
You may take this medication with or without food,